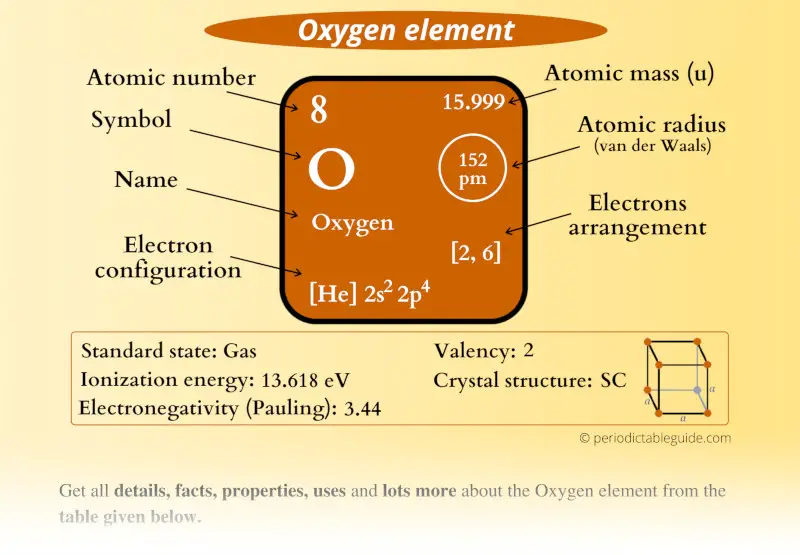
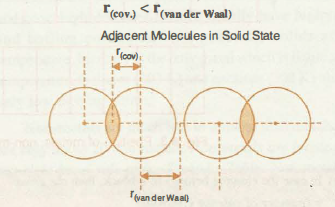
Revisiting van der Waals Radii: From Comprehensive Structural Analysis to Knowledge‐Based Classification of Interatomic Contacts - Chernyshov - 2020 - ChemPhysChem - Wiley Online Library

Table I from Quantum Mechanical Calculation of Molecular Radii. I. Hydrides of Elements of Periodic Groups IV through VII | Semantic Scholar
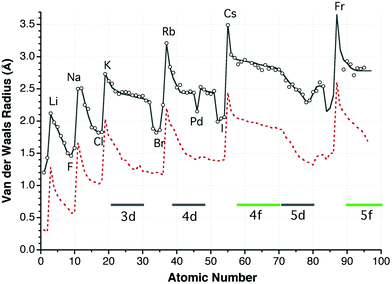
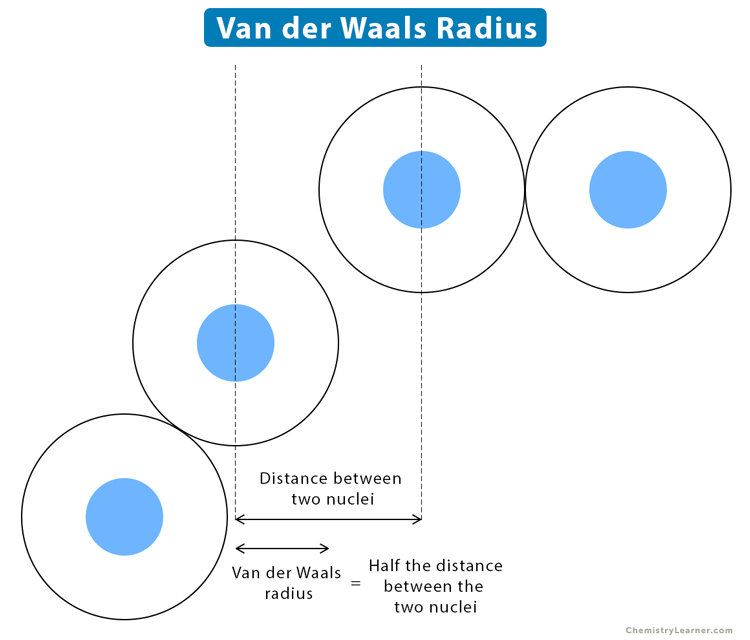
A cartography of the van der Waals territories - Dalton Transactions (RSC Publishing) DOI:10.1039/C3DT50599E

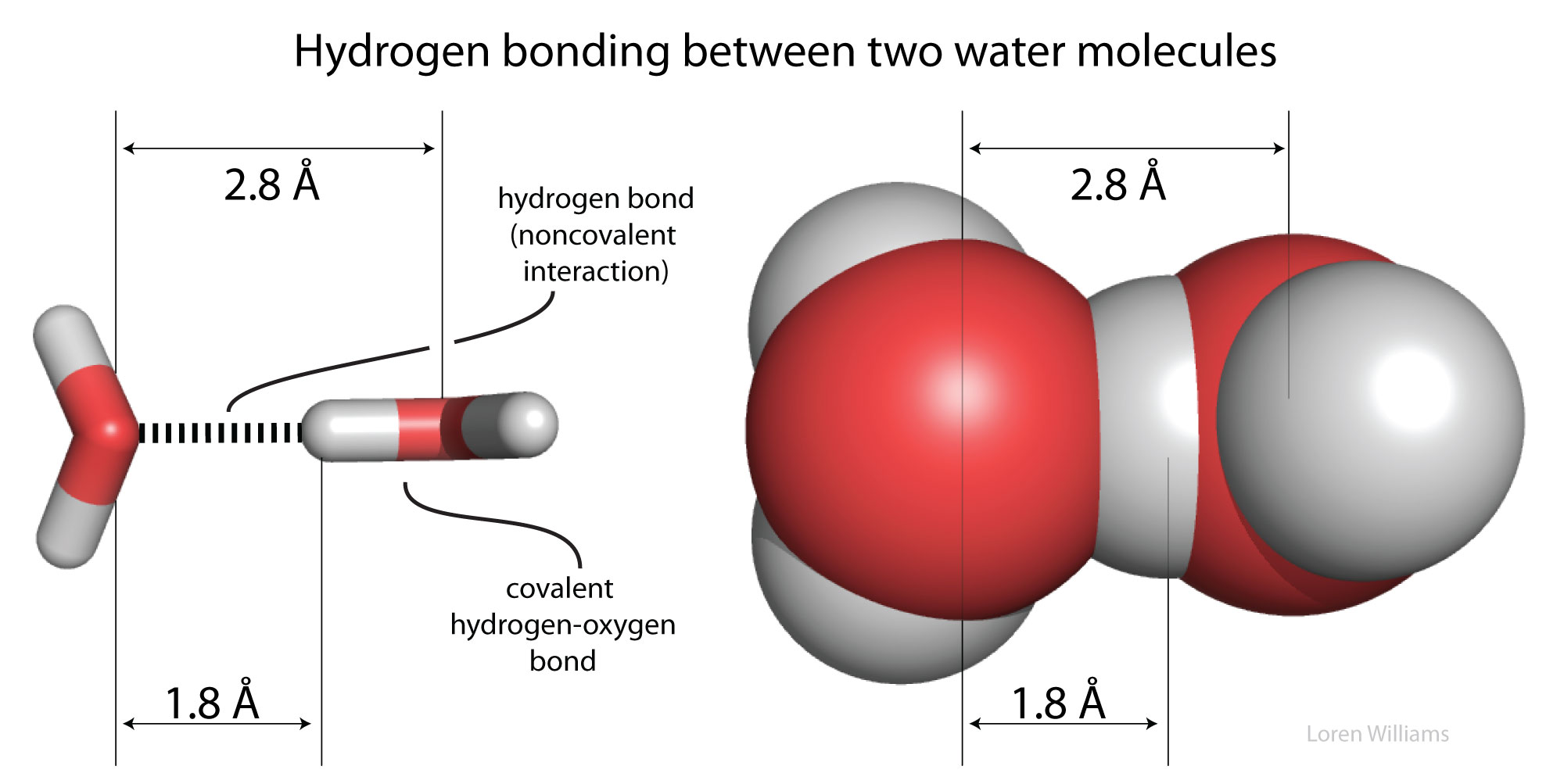
a. Give and explain the decreasing order of atomic radius of fluorine (F), nitrogen (N) and oxygen (O). b. Give the decreasing order of van der Waals radii: N, O, H, Cl,

Ozone Molecule Ball And Stick Model Stock Photo - Download Image Now - Ozone Layer, Molecule, Molecular Structure - iStock
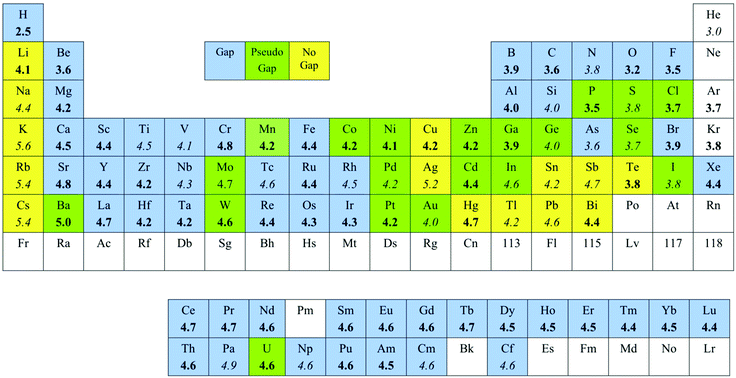
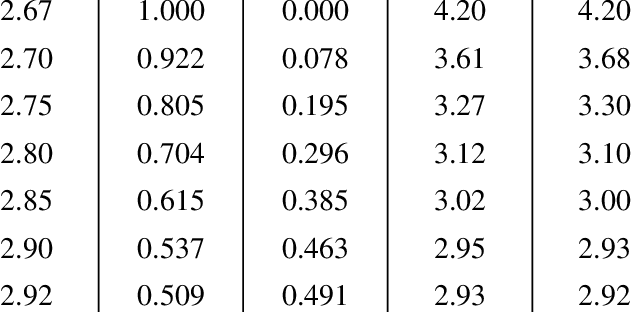
![PDF] Consistent approaches to van der Waals radii for the metallic elements | Semantic Scholar PDF] Consistent approaches to van der Waals radii for the metallic elements | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/7f2faf83943f15ccdb50e6b2992250dc4b784bef/5-Table2-1.png)